Understanding the pathology of neurodegenerative and psychiatric disorders caused by protein misfolding


Motomasa Tanaka, Ph.D.
Team Director, Protein Conformation Diseases
motomasa.tanaka@riken.jp
Research Overview
We are investigating the molecular basis of neurodegenerative disorders and psychiatric diseases using both in vivo and in vitro systems via a variety of techniques including neurobiology, structural biology, genetics and proteomics. In conjunction, we are seeking to uncover the structural basis of yeast prion strains and transmission barriers, and to identify novel functional prions or protein aggregates that play physiological roles in cells.
Main Research Fields
Biological Sciences
Related Research Fields
Chemistry / Complex Systems / Biological Sciences / Biology / Medicine, Dentistry & Pharmacy / Biology at molecular to cellular levels, and related fields / Biology at cellular to organismal levels, and related fields / Neuroscience and related fields
Keywords
- Neuropsychiatric and neurodegenerative disease
- Protein aggregation
- Amyloid
- mRNA translation
Selected Publications
- Endo R., Chen Y.K., Burke J., Takashima N., Suryawanshi N., Hui K.K., Miyazaki T., Tanaka M.:
"Dysregulation of ribosome-associated quality control elicits cognitive disorders via overaccumulation of TTC3."
Proc. Natl. Acad. Sci. U. S. A., 120, e2211522120 (2023).
10.1073/pnas.2211522120 - Nakagawa Y., Shen C-h,, Komi Y., Sugiyama S., Kurinomaru T., Tomabechi Y., Krayukhina E., Okamoto K., Yokoyama T., Shirouzu M., Uchiyama S., Inaba M., Niwa T., Sako Y., Taguchi H., Tanaka M.:
"Amyloid conformation-dependent disaggregation in a reconstituted yeast prion system."
Nat. Chem. Biol., 18, 321-331 (2022).
10.1038/s41589-021-00951-y - Shida T., Kamatari Y.O., Yoda T., Yamaguchi Y., Feig M., Ohhashi Y., Sugita Y., Kuwata K., and Tanaka M.:
"Short disordered protein segment regulates cross-species transmission of a yeast prion."
Nat. Chem. Biol. 16, 756-765 (2020) (cover).
10.1038/s41589-020-0516-y - Sugiyama, S., Tanaka, M.:
"Distinct segregation patterns of yeast cell-peripheral proteins uncovered by a method for protein segregatome analysis."
Proc Natl Acad Sci U. S. A., 116, 8909-8918 (2019).
10.1073/pnas.1819715116 - Hui, K.K., Takashima, N., Watanabe, A., Chater, T.E., Matsukawa, H., Nekooki-Machida, Y., Nilsson, P., Endo, R., Goda, Y., Saido, T.C., Yoshikawa, T., Tanaka., M.:
"GABARAPs dysfunction by autophagy deficiency in adolescent brain impairs GABAA receptor trafficking and social behavior."
Science Advances, 5(4), eaau8237 (2019).
10.1126/sciadv.aau8237 - Endo R., Takashima N., Nekooki-Machida Y., Komi Y., Hui K.K., Takao M., Akatsu H., Murayama S., Sawa A., and Tanaka M.:
"TDP-43 and DISC1 Co-Aggregation Disrupts Dendritic Local Translation and Mental Function in FTLD.
Biol. Psychiatry, 84, 509-521 (2018)."
10.1016/j.biopsych.2018.03.008 - Chen C.W., Tanaka M.
"Genome-Wide Translation Profiling by Ribosome-Bound tRNA Capture."
Cell Rep., 23, 608-621 (2018).
10.1016/j.celrep.2018.03.035 - Ohhashi Y., Yamaguchi Y., Kurahashi H., Kamatari Y.O., Sugiyama S., Uluca B., Piechatzek T., Komi Y., Shida T., Müller H., Hanashima S., Heise H., Kuwata K, and Tanaka M.
"Molecular basis for diversification of yeast prion strain conformation",
Proc. Natl. Acad. Sci. U. S. A., 115, 2389-2394 (2018).
10.1073/pnas.1715483115 - Tanaka, M., Ishizuka, K., Nekooki-Machida, Y., Endo, R., Takashima, N., Sasaki, H., Komi, Y., Gathercole, A., Huston, E., Ishii, K., Hui, K.K., Kurosawa, M., Kim, S.H., Nukina, N.,Takimoto, E., Houslay, M.D., and Sawa, A.:
"Aggregation of scaffolding protein DISC1 dysregulates phosphodiesterase 4 in Huntington's disease."
J. Clin. Invest., 127, 1438-1450 (2017).
10.1172/JCI85594 - Suzuki, G., Weissman, J.S., and Tanaka, M.:
"[KIL-d] protein element confers antiviral activity via catastrophic viral mutagenesis."
Mol. Cell, 60, 651-660 (2015).
10.1016/j.molcel.2015.10.020
News & Media
-
-

-
RIKEN research Fall 2023
"An over-reaction when protein synthesis stalls can lead to neurodevelopmental disorders in mice"
-
-
-
-
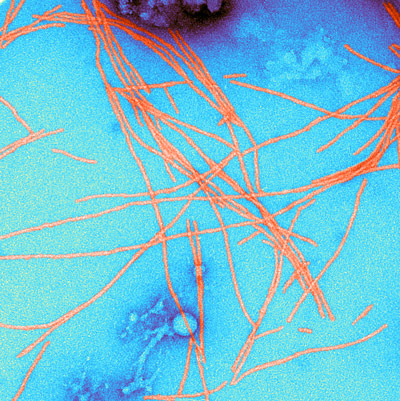
Three chaperones coordinate the breakup of amyloid fibrils in yeast
-
-
-
-
Protein pileup affects social behaviors through altered brain signaling
-
-
-

-
The benefits of misbehaving: prion helps yeast survive environmental stress
-
-
-

-
New yeast prion helps cells survive
-Prion proteins may be best known for their role in infectious diseases, but not all of them are destructive, new research shows-
-
-
-

-
Effect of yeast prion protein oligomer formation process on infection efficacy clarified
-
Lab Members
Principal investigator
- Motomasa Tanaka
- Team Director
Core members
- Ryo Endo
- Senior Research Scientist
- Takashi Nomura
- Research Scientist
- Audrey Marie Genevieve Ragagnin
- Research Scientist
- Yoshiko Nakagawa
- Postdoctoral Researcher
- Nayan Vinod Suryawanshi
- Postdoctoral Researcher
- Noriko Takashima
- Technical Staff I
- Shingo Tamai
- Postdoctoral Researcher
- Kai Sato
- Special Postdoctoral Researcher
- Reina Nakanishi
- Junior Research Associate
- Scott Jimmy Thomas
- International Program Associate
- Hideaki Takaku
- Technical Staff I
- Hiromi Kimura
- Technical Staff II
- Yuuki Tomita
- Technical Staff II Mail address
- Amrita Das
- Technical Staff II
- Yuxin Chen
- Research Part-time Worker I